What’s the difference between electronegativity and ionization energy?
Electronegativity is related to the pull of a nucleus on an electron being shared in a bond. Ionization energy is the energy required to remove an electron from an unbonded atom. Electronegativity is a ranking and has no units. Ionization energy is an amount of energy and has units such as kilojoules.
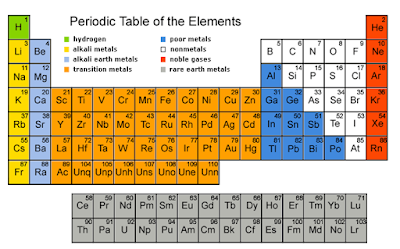
SPM Form 4: Periodic Table of elements (Checklist)
- Periodic Table: the table showing the elements in order of increasing proton number; similar elements are arranged in columns called groups.
- Group: A vertical column of elements in the Periodic table.
- Period: A horizontal row of the Periodic Table; its number tells you how many electron shells there are.
- Alkali metals: the Group I elements of the Periodic Table, which include lithium, sodium, potassium, rubidium, caesium and francium.
- Alkaline earth metals: the Group II elements of the Periodic Table, which include beryllium, magnesium, calcium, strontium, barium, and radium.
- Halogens: the Group VII elements of the Periodic Table, which include fluorine, chlorine, bromine, iodine and astatine.
- Noble gases: the Group 18 elements of the Periodic Table; they are called ‘noble’ or inert gases because they are so unreactive, which include helium, neon, argon, krypton, xenon and radon.
- Transition elements: the elements in the wide middle block of the Periodic Table (elements in group 3 to group 12).
- Metal: an element that shows metallic properties (for example conducts electricity, and forms positive ions)
- Non-metal - an element that does not show metallic properties: the non-metals lie to the right of the zig-zag line in the Periodic Table.
- Amphoteric oxide: An oxide that exhibits both acidic and basic properties.
- The atomic radius is a term used to describe the size of the atom.
- The ionization energy is the energy required to completely remove an electron from a gaseous atom or ion.
- Electron affinity reflects the ability of an atom to accept an electron. It is the energy change that occurs when an electron is added to a gaseous atom.
- Electronegativity is a measure of the attraction of an atom for the electrons in a chemical bond. The higher the electronegativity of an atom, the greater its attraction for bonding electrons.
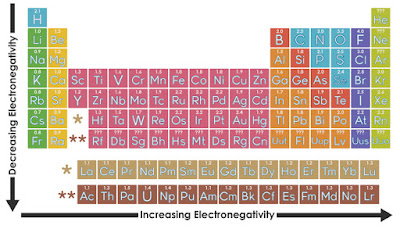
Electronegativity and Ionization Energy
Electronegativity
is related to ionization energy. Electrons with low ionization energies
have low electronegativities because their nuclei do not exert a strong
attractive force on electrons. Elements with high ionization
energies have high electronegativities due to the strong pull exerted on
electrons by the nucleus.
Electronegativity and Periodic Table Trends
Electronegativity is a
measure of the attraction of an atom for the electrons in a chemical bond. The
higher the electronegativity of an atom, the greater its attraction
for bonding electrons. In an element group, the
electronegativity decreases as atomic number increases, as a result of
increased distance between the valence electron and nucleus (greater
atomic radius). An example of an electropositive (i.e., low
electronegativity) element is cesium; an example of a
highly electronegative element is fluorine.
- Moving left to
right across the periodic table, electronegativity increases.
- Moving top to bottom down
the periodic table, electronegativity decreases.